A fetal-like repair mode may help the gut heal — and help explain why chronic inflammation can turn dangerous
A fetal-like repair mode may help the gut heal — and help explain why chronic inflammation can turn dangerous
Medicine often treats regeneration and cancer as opposing forces. On one side is the body trying to repair damaged tissue. On the other is tissue growing when it should not. But biology is rarely that tidy. Sometimes the very programmes that help tissue heal can, if they persist too long, edge towards something much more dangerous.
That is the grey zone a new intestinal regeneration study begins to illuminate. The research suggests that after inflammatory injury, intestinal tissue can temporarily shift into a fetal-like regenerative state. That partial return to a more primitive repair programme does not appear to be inherently abnormal. In fact, it may be part of how healing is supposed to work. The problem begins when the programme fails to switch off.
That is the most important idea in this new view of “fetal reversion” in the gut. The study does not argue that a fetal-like state is automatically pathological. It suggests something more subtle — and more interesting. This repair mode may be beneficial in the short term, but if it persists, especially when p53 signalling is lost, it may help explain chronic inflammation, IBD-related risk and progression towards colorectal cancer.
The intestine may have an emergency repair setting
The intestine is one of the most dynamic tissues in the body. Its lining constantly renews itself while facing mechanical, chemical, microbial and inflammatory stress. To maintain that balance, it relies on a tightly regulated regenerative system centred on the intestinal crypts and the cells that keep the epithelium replenished.
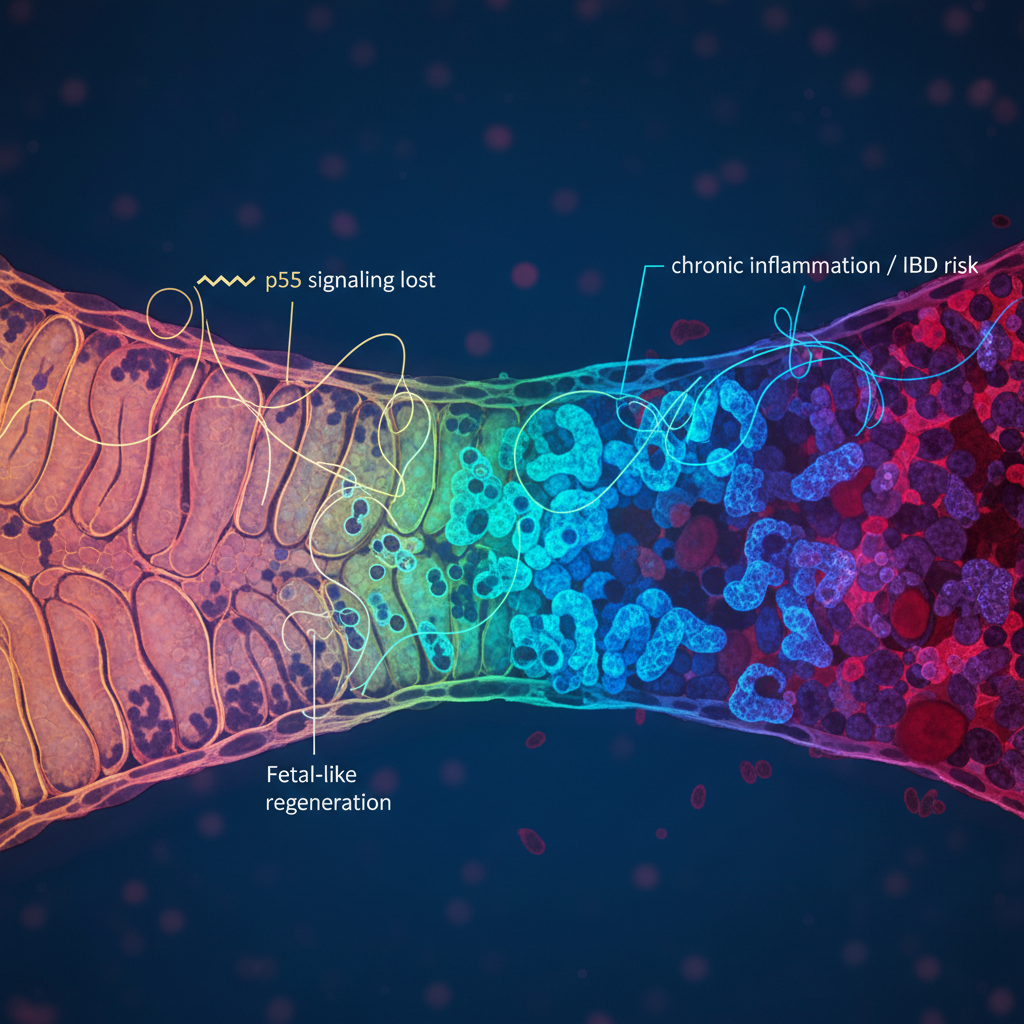
But when damage becomes severe, ordinary regeneration may not be enough. That is where the idea of fetal-like reversion comes in. Instead of simply replacing lost cells using the usual adult programme, injured tissue appears capable of shifting into a more plastic, proliferative and development-like state — one that resembles aspects of fetal tissue.
Biologically, that makes sense. Fetal tissues are built for rapid growth and structural reorganisation. In the context of acute damage, temporarily recovering some of that plasticity could be an efficient way to rebuild the epithelium.
What the study found
The supplied study directly supports that interpretation. Following colitis-related injury, the intestinal epithelium entered a transient fetal-like regenerative state. At the same time, p53 signalling was strongly activated.
That detail matters. In normal tissue, the study suggests p53 is not merely responding to stress in a general way. It appears to help terminate the regenerative programme once repair has progressed far enough.
In other words, the fetal-like state may be part of normal healing — but only if the tissue can exit it at the right time.
When p53 was absent, the story changed. Intestinal crypts remained locked in a highly proliferative regenerative state over the long term. Rather than using the programme briefly and then returning to homeostasis, the tissue stayed trapped in a mode of abnormal growth.
That provides a plausible mechanistic bridge between inflammatory injury, failed resolution of repair and increased cancer risk.
p53 may be doing more than tumour suppression
p53 is already well known as one of the body’s central tumour suppressors. It helps control cell-cycle arrest, DNA damage responses, apoptosis and other safeguards against uncontrolled growth. The new study suggests it may also have a more specialised role in injured intestinal tissue: helping to shut down an otherwise useful repair programme.
That is an important shift in perspective. The danger may not simply be that regeneration happens, but that regeneration does not know when to stop.
In a tissue as proliferative as the gut, that distinction matters. A growth-heavy repair programme may be beneficial for days or weeks after injury. But if it remains active for too long, it may begin to favour disorganisation, persistent proliferation and a tissue environment more permissive to malignant transformation.
Why this matters in inflammatory bowel disease
This idea is especially relevant in inflammatory bowel disease. In conditions such as ulcerative colitis and Crohn’s disease, the intestine can cycle repeatedly through injury, inflammation and repair. Over time, that chronic damage is known to increase the risk of colorectal cancer.
The new work offers a plausible explanation for part of that link. If inflammatory injury repeatedly activates a fetal-like repair state, and if successful healing depends on switching that programme off, then it makes sense that repeated injury — or failure of key regulatory pathways — could leave tissue stuck in a state of prolonged abnormal regeneration.
That does not mean fetal-like reprogramming explains all cancer risk in IBD. The study does not support such an expansive claim. But it does suggest that one part of the danger may lie not only in the inflammatory damage itself, but in what happens when the repair response fails to resolve properly.
Regeneration and cancer may share more biology than we like to admit
One of the most striking implications of this work is how much regeneration and cancer seem to borrow from the same biological language. Both involve proliferation, tissue remodelling, cellular plasticity and activation of programmes that, in other contexts, resemble development.
That does not mean regeneration is simply early cancer. But it does suggest the body uses some of the same tools for healing and for growth. What separates a helpful response from a dangerous one is often duration, control and context.
In the intestine, the study suggests fetal-like reprogramming may be one of those tools. Helpful when used briefly, hazardous when allowed to persist.
A different way to think about colorectal cancer risk
Perhaps the most important contribution of this work is conceptual. It shifts the model of IBD-related colorectal cancer away from a purely damage-based story.
Chronic inflammation still matters enormously. But the new findings suggest that the quality of tissue repair may matter too. The risk may not depend only on how often the tissue is injured, but on how effectively it exits repair mode afterwards.
A gut that repairs itself and restores normal epithelial identity may return to balance. A gut that remains locked in a proliferative, fetal-like state may accumulate instability and abnormal growth pressures over time.
That is a useful refinement because it frames cancer risk not only as a consequence of repeated injury, but also as a consequence of incomplete resolution.
What this does not prove
It is important to stay proportionate.
The evidence comes from a single mechanistic study, mainly using murine and organoid models. That makes it valuable for biological explanation, but still a long way from direct clinical application.
This study does not establish a new treatment for IBD. It does not offer a validated strategy for colorectal cancer prevention in patients. And it does not imply that fetal-like reversion is inherently harmful. In fact, the study supports the opposite view: this state may be a normal and necessary part of repair.
The problem, at least based on the current evidence, seems to be persistent dysregulation rather than the regenerative state itself.
Where this line of research may lead
Even so, the work opens important questions. Could clinicians eventually identify patients whose intestinal repair remains abnormally prolonged? Might biomarkers of this fetal-like regenerative state help flag higher cancer risk in IBD? Could future therapies help tissue exit this emergency programme more cleanly without impairing healing?
Those possibilities remain speculative. But the value of this study lies in making the questions more concrete.
The most balanced takeaway
The supplied research supports the idea that intestinal injury can trigger a transient fetal-like regenerative state, and that p53 plays an important role in shutting that programme down once repair is underway. When that brake is lost, intestinal crypts can remain trapped in a highly proliferative state, offering a plausible mechanistic link between chronic inflammation, abnormal regeneration and colorectal cancer risk.
The key message is not that fetal-like reversion is inherently bad. It may be part of normal healing. The problem appears to arise when the intestine cannot leave that emergency mode.
That makes the story of inflammation-related cancer more nuanced — and probably more biologically realistic. In some cases, what threatens the gut may be not only the injury it suffers, but the way it tries to heal and cannot fully stop.