How the body holds ‘sleeping’ tuberculosis in check: scientists are clarifying the role of granulomas and immune defence
How the body holds ‘sleeping’ tuberculosis in check: scientists are clarifying the role of granulomas and immune defence
The phrase “sleeping tuberculosis” is useful shorthand, but it hides a much more complicated biological reality. When researchers talk about latent tuberculosis, they are not necessarily describing bacteria that are fully inactive, sealed away and permanently silent. What science increasingly suggests is something more dynamic: a long-running contest in which the immune system manages to contain infection without fully eradicating it.
That balance is exactly what researchers are trying to understand more clearly. The line of work highlighted here points to a more detailed picture of how the body “traps” tuberculosis through immune mechanisms and structures known as granulomas. The central idea is well supported by the supplied material: latent tuberculosis depends on active host containment, not simply on bacteria passively going dormant.
At the same time, the evidence should not be overstated. What is available here says more about mechanisms and models than about one definitive, fully mapped process in living humans.
Latent tuberculosis is not just an infection in pause mode
For years, latent tuberculosis was often imagined almost like a waiting room state: the bacterium remained hidden and inactive until some breakdown in immunity allowed it to reactivate. That view is still partly useful, but it is no longer enough.
More recent reviews suggest that latent TB biology may work more like a spectrum. Instead of a single bacterial condition, there may be different degrees of activity, persistence and immune control. That means the idea of “sleeping TB” as one static, fully dormant state probably oversimplifies what is happening.
This matters because it changes the scientific question. Instead of asking only why the bacterium “wakes up”, researchers are increasingly asking how the body keeps it under control for so long — and why that control sometimes fails.
Granulomas sit at the centre of the story
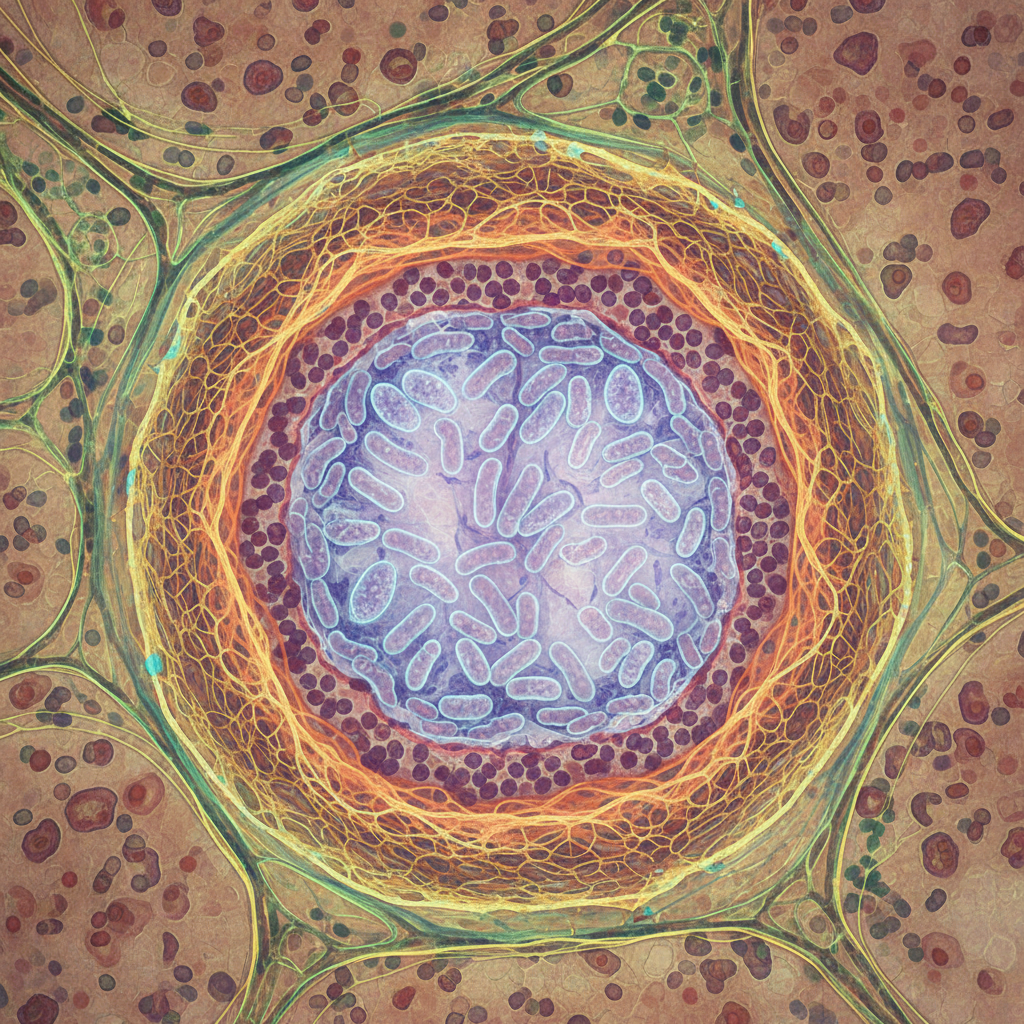
At the centre of this picture are granulomas, organized structures made up of immune cells at the site of infection. In simple terms, they are the body’s attempt to isolate something it cannot completely eliminate.
In tuberculosis, the granuloma acts as a form of biological containment. It is not just a scar or a static clump of cells. It is a living structure shaped by inflammatory signalling, cellular immune responses, tissue remodelling and the persistent presence of the bacterium itself.
That is one reason the language of “trapping” makes sense, as long as it is used carefully. The body really does try to wall off and restrict Mycobacterium tuberculosis. But it does not do so in an absolute or necessarily permanent way. The granuloma represents containment, not final victory.
Which immune defences help maintain that balance
The supplied reviews reinforce that several immune pathways are important in keeping tuberculosis under control. Among the most prominent are T cells, TNF-alpha and interferon-gamma.
These are part of the cellular immune response that helps recognize infection, activate macrophages and sustain the inflammatory environment needed to restrict bacterial spread. Without that coordinated response, containment becomes more fragile and the risk of progression to active disease rises.
This helps explain why people with immune suppression, especially those with defects in key cellular immune pathways, are at increased risk of TB reactivation. What breaks down is not just a general barrier, but a more specific immune architecture that had been helping preserve the balance between bacterial persistence and host control.
Containment is not the same as elimination
One of the most important messages from this field is that the body can control tuberculosis without completely clearing it. That may be the single most important difference between latent TB and many acute infections.
In practice, this means the immune system may limit bacterial expansion, prevent dissemination and keep infection clinically silent for years. But that control may remain incomplete. Some bacteria may persist in protected niches, possibly in varying metabolic states, waiting for an opportunity to expand again.
That view is less tidy than the image of bacteria simply “sleeping”, but it is probably closer to reality. Latency appears to be less like a full pause and more like an uneasy truce.
What calcification adds to the picture
One of the supplied articles focuses on calcification in granulomas and the complications associated with it. That does not by itself prove how the body contains latent infection, but it does help show what can happen during the chronic evolution of these structures.
Calcification may represent one possible outcome of long-standing, persistent granulomas. In some cases, it suggests a prolonged attempt by the body to stabilize and encapsulate the lesion. That reinforces the idea that granulomas are not brief immune events but part of a longer remodelling process.
Still, it is important not to confuse that with direct proof of the full biology of latent containment. Calcification is one clue within granuloma biology, not a complete explanation of how latent TB is held in check.
Why experimental models matter
Another useful part of the supplied material is a human in vitro granuloma model. According to the cited study, latent tuberculosis infection can generate granuloma-like aggregates with characteristic immune responses, offering a tool for studying early containment mechanisms.
That matters because it gives researchers a controlled way to observe how human cells organize a response resembling the kind believed to occur during early infection containment. Instead of relying only on indirect evidence or clinical inference, scientists gain a system for testing more specific mechanistic questions.
But the limitations are equally important. A granuloma model grown in the lab cannot fully reproduce the complexity of latent tuberculosis in a living lung or in the broader environment of human tissues. It is a useful mechanistic tool, not a substitute for the disease itself.
What it really means to “map” latent TB
The headline suggests scientists are mapping how the body traps tuberculosis. That is fair if “mapping” means identifying key pathways, cells, structures and interactions. In that sense, the evidence does support a more detailed picture of immune containment.
But it would be too strong to take that as meaning there is now one complete, closed map of latent TB in humans. The supplied literature includes reviews, mechanistic studies, work on granulomas and experimental models. That is valuable, but it is not the same as direct in vivo mapping of every aspect of latent infection in human tissues.
What is emerging is a better understanding of a dynamic system: persistent bacteria, structured tissue responses, evolving granulomas and an immune system trying to hold the balance.
Why this matters for public health
Tuberculosis remains one of the most important infectious diseases in the world, and latent infection is central to that burden. Millions of people may carry the bacterium without symptoms, with varying risks of progressing to active disease.
Understanding immune containment better matters because it could eventually help researchers distinguish more clearly between stable latency and states closer to reactivation. It could also shape future thinking about biomarkers, vaccines and prevention strategies.
But that future-facing possibility should be framed carefully. The supplied evidence does not show an immediate change in patient care. The advance is in disease biology, not in a ready-to-use clinical application.
The most balanced reading
Taken together, the supplied studies support the idea that latent tuberculosis depends on complex host mechanisms of containment, not simply on bacteria entering a fully dormant state. Granulomas appear central to that process, while immune pathways involving T cells, TNF-alpha and interferon-gamma help maintain control. Human granuloma models in the lab also offer useful tools for studying how this containment may begin and evolve.
At the same time, the evidence remains more mechanistic and model-based than a definitive map of latent TB in living human tissues. The phrase “sleeping tuberculosis” oversimplifies a biology that likely involves multiple bacterial states rather than one fixed dormant condition.
The safest conclusion, then, is this: scientists are building a clearer picture of how the body contains latent tuberculosis through immune defence and granuloma biology. That is an important advance in understanding the disease, but it does not mean the mechanism has been fully solved, nor that patient care changes immediately as a result.