Why smoking may raise dementia risk: a new theory links the lungs, exosomes, and brain iron — but the evidence still needs caution
Why smoking may raise dementia risk: a new theory links the lungs, exosomes, and brain iron — but the evidence still needs caution
Smoking has long been understood as a major threat to the lungs, heart, and blood vessels. But over time, it has also become clear that tobacco exposure is bad for the brain. The most familiar explanation is vascular: smoking damages blood vessels, increases inflammation, raises stroke risk, and impairs circulation, all of which can contribute to worse cognitive aging.
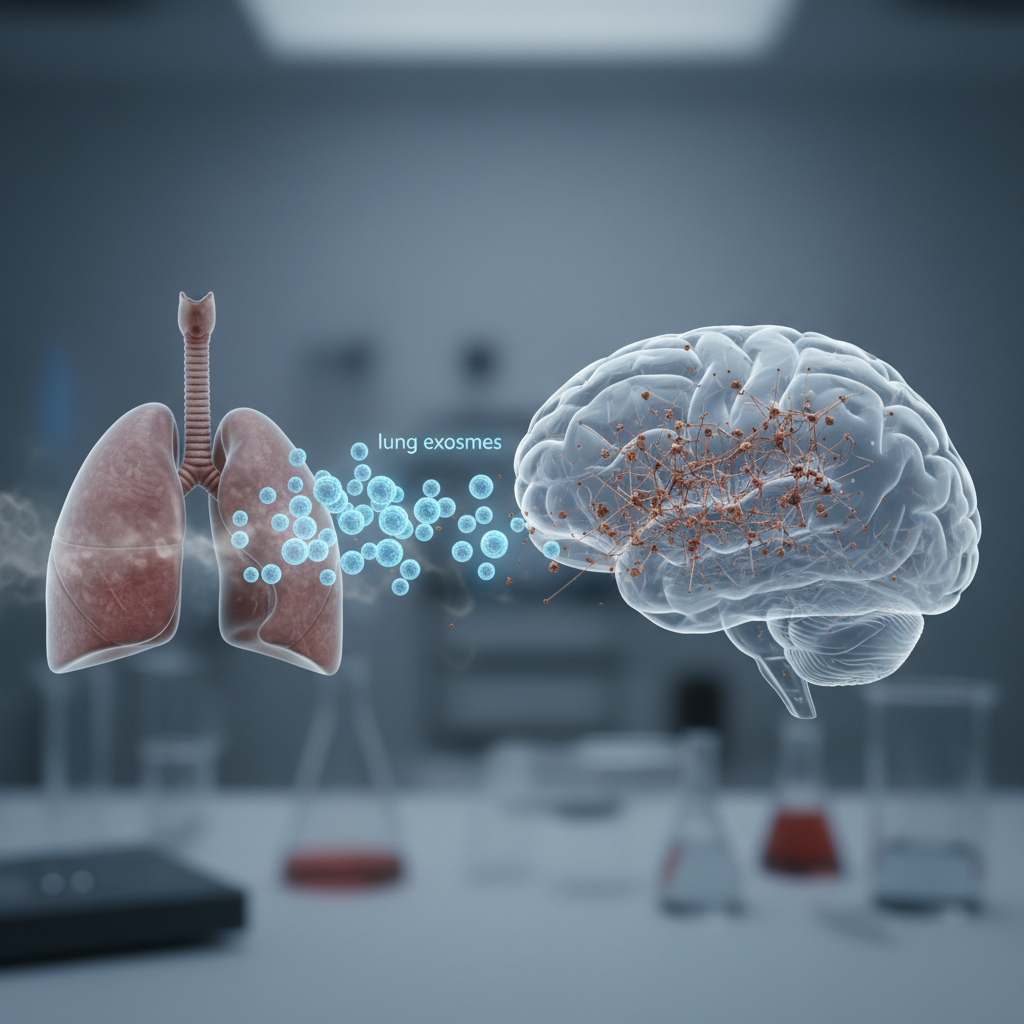
The new headline points to a more specific and more intriguing possibility. Rather than focusing only on classic vascular damage, it suggests that the lungs may send biological signals to the brain through exosomes — tiny vesicles released by cells that carry proteins, lipids, and genetic material. According to the proposed mechanism, those lung-derived signals could disturb brain iron balance, potentially contributing to dementia risk.
It is an interesting idea. It is also one that needs to be handled with considerable caution.
What the headline is really suggesting
The logic appears to be this: smoking injures lung tissue and changes the way the lungs communicate with the rest of the body. Part of that communication may occur through exosomes released into circulation. If those exosomes reach the brain, they might interfere with iron regulation there.
That matters because iron is essential to brain function, but it is also biologically delicate. When iron is present in the wrong place, in the wrong amount, or in a poorly regulated state, it can contribute to oxidative stress, inflammation, and cellular damage.
If this mechanism is confirmed, it could offer a more detailed explanation for smoking and dementia risk. Instead of relying only on the well-known story of vascular injury, researchers might begin to view smoking-related brain damage partly through a lung-to-brain signalling pathway as well.
That idea is plausible within modern biology. Organs do not operate in isolation. They communicate constantly, and that communication is increasingly being studied in chronic disease, including neurodegeneration.
Why iron matters in brain disease
Iron plays a necessary role in the brain. It is involved in energy production, cellular metabolism, and several processes that neurons rely on to function properly. But iron is also potentially harmful if its regulation breaks down.
That is one reason iron keeps appearing in discussions of neurodegenerative disease. In conditions such as Alzheimer’s disease and Parkinson’s disease, researchers have shown growing interest in whether altered iron handling may contribute to brain injury.
So a hypothesis linking smoking, lung-derived exosomes, and disrupted iron balance in the brain does not sound biologically far-fetched. In fact, it fits fairly well with current interest in systemic drivers of brain disease and the possibility that signals originating outside the brain may shape neurodegeneration.
The biggest problem: the mechanism could not be independently verified
This is the most important caution in the story. No PubMed studies were supplied to independently support the specific claim in the headline.
That creates a major limitation. Without the underlying scientific papers, it is not possible to assess several basic questions, including:
- whether the evidence comes from cell models;
- animal studies;
- human tissue or human samples;
- or epidemiological data.
It is also impossible to tell whether this is an early preclinical finding, a more mature translational result, or simply a mechanistic idea still at a very preliminary stage.
That lack of verifiable evidence changes how the story should be read. The mechanism may turn out to be real and important. But with the material supplied here, it cannot be treated as independently confirmed.
Why the idea still fits current science
Even though the specific claim cannot be verified from the supplied evidence, the broader concept fits with several active research trends.
One is the growing interest in cross-organ communication. Researchers increasingly study how the gut, liver, lungs, immune system, and adipose tissue may influence the brain through circulating signals. Exosomes and other extracellular vesicles are part of that conversation because they are one way cells and organs exchange biological information.
In that context, a lung-to-brain signalling pathway triggered by tobacco exposure would not be surprising. Smoking causes widespread inflammation, oxidative stress, toxic exposure, and metabolic disruption. It would make sense if some of its effects on the brain were mediated through circulating molecular messages rather than only through local lung injury or blood vessel damage.
Another relevant theme is the role of iron homeostasis in neurodegeneration. Because disturbed iron handling has already drawn attention in brain disease research, the idea that smoking could affect dementia risk through iron imbalance is scientifically coherent, even if not yet proven in this specific form.
What should not be concluded
Because the evidence here is weak, several stronger claims should be avoided.
The first is that this pathway has been proven in humans. It has not been independently verified from the supplied material.
The second is that this would be the main explanation for why smoking raises dementia risk. Even if the mechanism is eventually confirmed, it would probably be one part of a much larger picture that includes:
- vascular damage;
- systemic inflammation;
- oxidative stress;
- metabolic effects;
- and possibly other pathways still being worked out.
The third is that this finding changes prevention or treatment in immediate practical ways. Nothing in the supplied evidence supports that kind of leap.
What this hypothesis adds to the smoking-and-brain story
Even with those limitations, the headline adds something important. For years, public discussion of smoking and brain health has been dominated by indirect explanations: worse circulation, higher cardiovascular risk, more stroke, and faster overall aging. Those explanations remain valid.
But mechanisms like the one proposed here suggest tobacco may not be only an indirect brain toxin. It may also be involved in wider biological signalling networks that affect the nervous system through inflammation, cell-to-cell communication, and molecular imbalance.
If that kind of pathway is eventually confirmed, it could make the smoking-dementia link more biologically detailed and more convincing. That matters because dementia is unlikely to arise from any single cause. It probably reflects the cumulative effects of vascular, metabolic, inflammatory, and neurobiological injury over time.
Why a weak-mechanism story can still matter
Not every important biomedical finding arrives fully ready for clinical use. Some matter because they reshape the questions researchers ask.
In this case, the proposed lung-exosome-brain-iron pathway raises useful questions:
- Can the lungs send neurotoxic signals in response to smoking?
- Could those signals help explain part of smoking’s cognitive risk beyond vascular injury?
- Is altered brain iron a cause, a downstream consequence, or a marker of damage?
- Would this matter only in laboratory models, or also in people?
These questions remain unanswered. But they are scientifically worthwhile, and that gives the hypothesis value even before it is fully established.
What readers should take from this
For the public, the safest takeaway is not “scientists have now explained why smoking causes dementia”. That would go too far.
A better reading is this: researchers may have identified a plausible biological pathway linking smoking to dementia risk through lung-derived signalling and disturbed brain iron balance, but the specific claim could not be independently verified from the supplied evidence.
That does not weaken concern about smoking. If anything, it reinforces the broader point that tobacco’s effects on the brain may be more complex than the traditional vascular story alone suggests. Even without this specific mechanism being confirmed, there is already strong reason to take smoking’s long-term brain effects seriously.
The most balanced conclusion
The most responsible interpretation is that this is a plausible but unverified mechanistic story. A lung-to-brain pathway involving exosomes and altered iron regulation would fit with current scientific interest in cross-organ signalling and neurodegeneration. If confirmed, it could help explain how smoking contributes to dementia risk through more than vascular damage alone.
But the evidence provided here is weak for validating the headline’s specific claim. No PubMed articles were supplied for independent verification, so it is not possible to judge whether the mechanism rests on cell studies, animal work, human samples, or population data. It is also unclear how early-stage the finding may be.
So the strongest safe conclusion is this: the proposed pathway is biologically interesting and worth watching, but it should not be treated as a proven explanation for smoking-related dementia risk. For now, it is best understood as a promising hypothesis that may add to the broader picture of how tobacco harms brain health.